Better Dispersants for Minerals
Problem
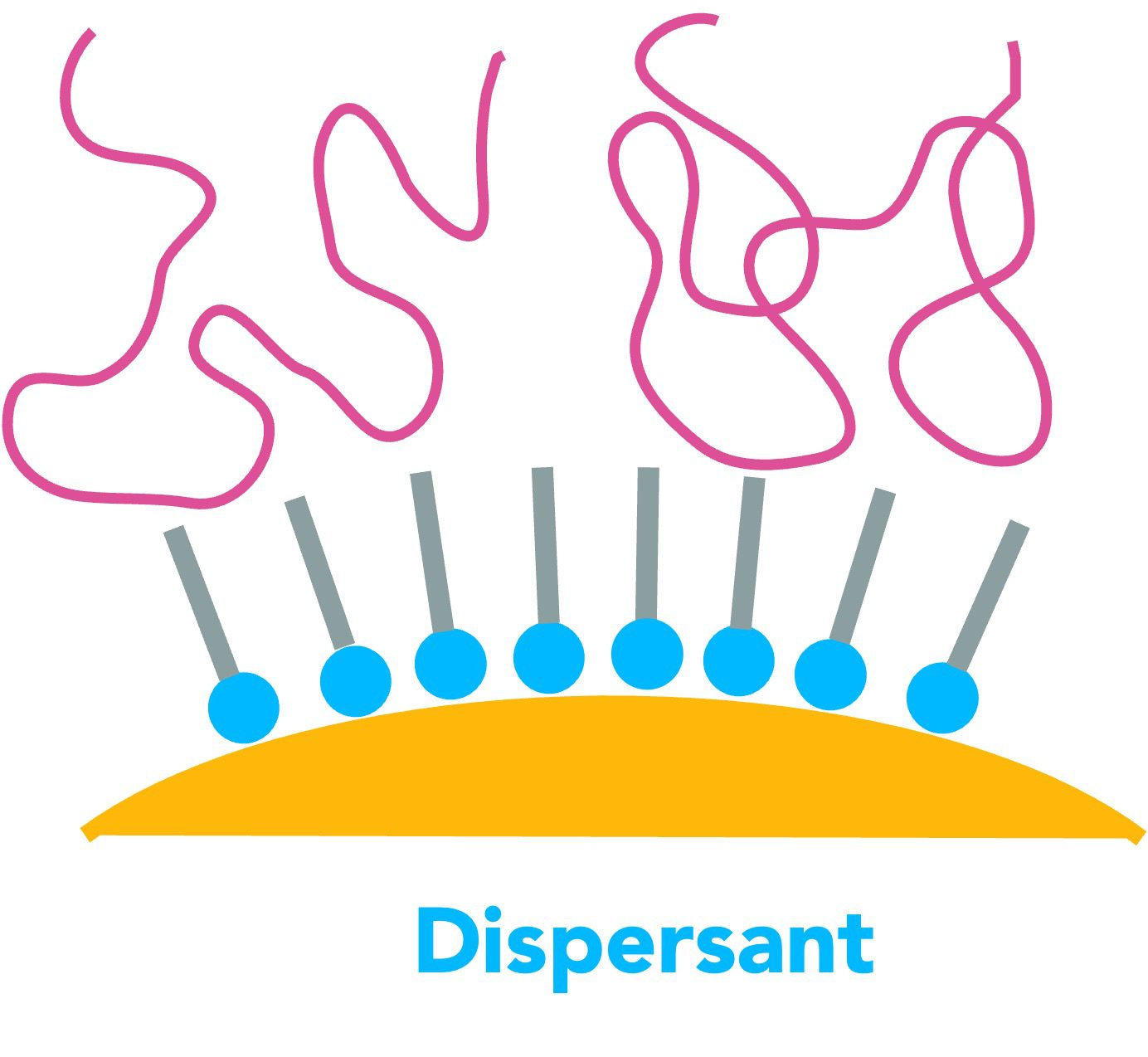
In many industrial products, a high loading of mineral filler is needed. For example, halogen free flame retardant minerals like ATH, MDH or HMH are only effective when used at 60 weight percent concentration or higher. PVC floor tiles can have 80% or more of calcium carbonate filler in order to decrease cost. Even household appliance parts are routinely made from polypropylene filled with 40-60% filler by weight. However, high loadings can cause poor flow, decreased gloss, reduction in strength and a dramatic sacrifice in impact resistance and elongation to break. In this particular instance, the customer was a long-standing client of mine. They manufactured high performance dispersants and coupling agents but their normal range of products did not perform well enough in a magnesium hydroxide filled EVA compound for cable jackets. They turned to Phantom for ideas on how to make better dispersants for minerals and take performance to the next level.
Action
My client called me and I still remember it to this day because I was on holiday standing next to a swimming pool. Despite being on vacation, I like to be there for my retainer clients whenever they need me, so I took the call. They said that they had tried the usual dispersants in their portfolio but the loss in mechanical properties of the cable jacket was too great. Knowing their product range well, I had to agree that they had already tried the best alternatives. It was unclear what more we could do to help their customer meet the target specifications.
Solution
As the normal products and chemistries had not been good enough, it seemed that the solution would have to be something really new. I hesitated to suggest what I had in mind because it can be expensive and time-consuming to create new products. Nevertheless, it was my duty to provide any ideas that might work to my client. So, I described on the phone what the “ideal” structure of dispersant chemical would be, i.e. a structure that I thought likely to perform even better than any products on the market. I braced myself for a negative response. I expected them to say that they could not possibly create such a new additive. Instead of that reaction, I got a pleasant and shocking response. They said:
“We already synthesized that molecule years ago and have a sample of the shelf!”
They had made the molecule in their R&D department but never found a use for it, so it had just sat on the shelf for years. They arranged to perform new experiments with the MDH filler and were astounded at the performance. The mechanical performance, especially the elongation to break was by far the best they had ever seen. Even now, years later when that person and I meet at a trade show, we reminisce over this great collaboration.
This is another illustration of how the right advice can save huge amounts of time and money. Because I had many years of experience designing dispersants and coupling agents staring in with my Master’s and PhD studies, it was possible to imagine the solution and get great results right away. As a wise person once said “the more I practice, the luckier I get”.
More information on better dispersants for minerals can be found on this page.
More specifics on flame retardant fillers and other functional fillers can be found on this page.
Some excellent books on this topic include:
Practical Dispersion: A Guide to Understanding and Formulating Slurries, 3rd Edition
