PLASTICS & THE ENVIRONMENT
PROPOSED DBDPE BAN
evidence & perspective from the leading INDEPENDENT expert
A DBDPE Ban has been Proposed –
What are the Facts?
The Canadian government has recently announced a proposal to ban DBDPE, a flame retardant used to save lives. Is this a wise move to protect the environment, or is it a mistake likely to endanger lives? The only way to know is to check the science.
This article looks covers all of the details about the proposed ban on DBDPE.
- What is DBDPE?
- What does DBDPE do?
- Is DBDPE safe?
- What is the basis for the proposed ban of DBDPE?
- Is the proposed DBDPE ban justified?
Let’s look at the peer-reviewed science to find the answers.
This page is based on the official objection letter – download here.

What are Brominated Flame Retardants?
Fires have caused damage, injury and death to humans for all of recorded history. Flame retardants were invented to prevent combustion, and to slow the spread of fire, so that people have more time to escape. Their use is mandated by law and proven to save thousands of lives. as well as hundreds of millions in property damage per year.
There are many types of flame retardant additives, and each has pros and cons. Brominated flame retardants are among the most effective. If 10% brominated flame retardant is effective, then up to 60 percent by weight of a mineral-based flame retardant, like ATH, HMH or MDH, would be needed to do the same job. Adding 60 percent by weight is not optimal, and severe drawbacks in mechanical properties will result.
Flame retardants are vital additives that are proven to save thousands of lives every year in Canada and around the world. There are 7000 residential fires per year in Canada, plus, of course, industrial fires. Approximately 70 injuries and 11 deaths occur per 1000 fires. The data shows that the very young and the elderly are most at risk and experience far higher death rates in fire situations.
Clare and H. Kelley Roe, Fire and at risk populations in Canada Analysis of the Canadian National Fire Information Database, University of the Fraser Valley – Center for Public Safety & Criminal Research, 2017
Toxicological Risks of Selected Flame-Retardant Chemicals, National Academy of Sciences, 2000
“In this study the most fire retarded furniture burned more slowly and produced less acutely toxic smoke and less total smoke than the furnishings with poor protection from fire.”
“This affords as much as 5 times the escape time for a fire in this configuration as well as time for the fire department/ brigade to respond. Based on fire department response standards in the US, it is likely that a fire with this much delay could be extinguished before the structure is lost as well as allowing additional time for occupants to evacuate.”
M. S. Blais et al., Comparative Room Burn Study of Furnished Rooms from the United Kingdom, France and the United States, Fire Technology, 56 489-514, 2020
“…late 2009 The Department for Business, Innovation and Skills (BIS) published a report comparing the number of fires, injuries and deaths attributable to Furniture and Furnishings for the periods 1981-5 and 2002-7. Overall, in the period 2002-07, the analysis suggests that the FFRs account for:
54 fewer deaths per year
780 fewer non-fatal casualties per year
1,065 fewer fires each year”
https://blog.polymers.co.uk/flame-retardants-save-lives
Since the introduction of flame retardant additives, mortalities have halved.
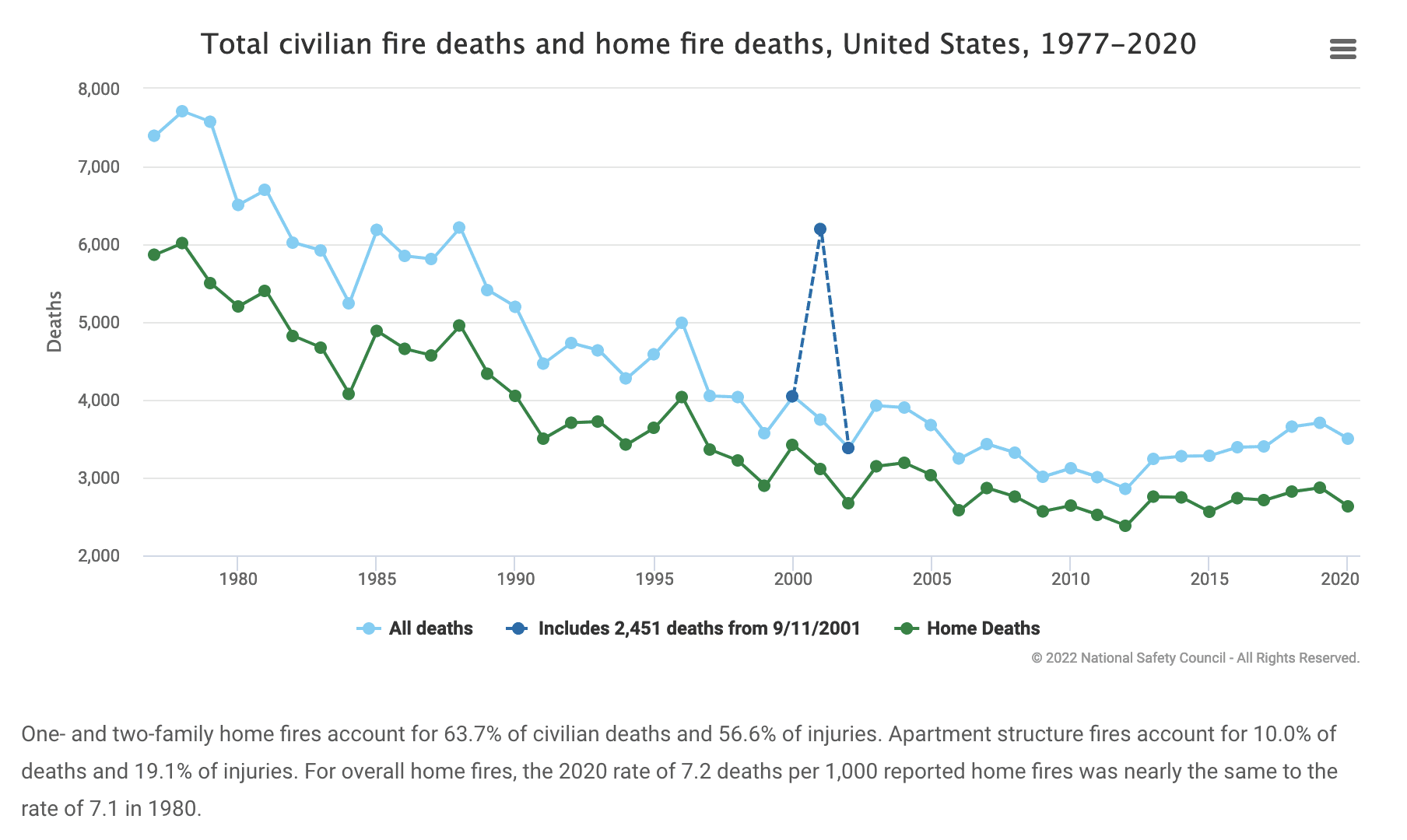
National Safety Council – Injury Facts https://injuryfacts.nsc.org/home-and-community/safety-topics/fire-related-fatalities-and-injuries/
What is DBDPE Flame Retardant?
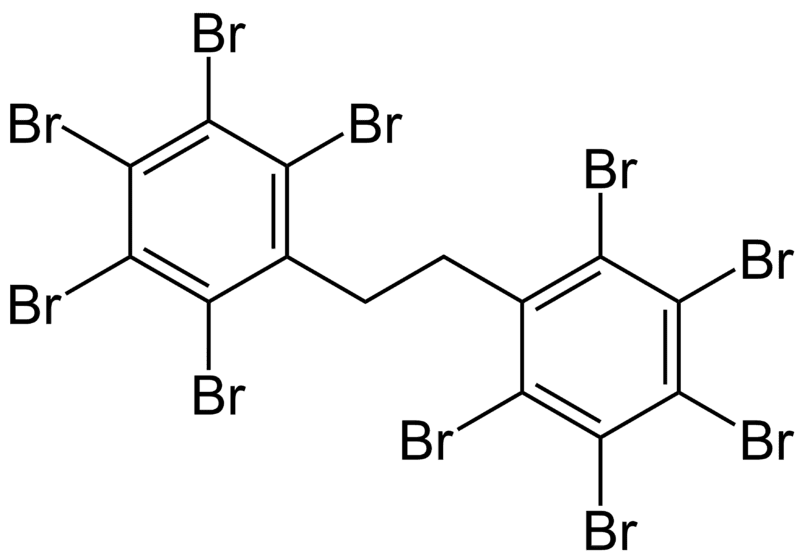
The diagram shows the chemical structure of DBDPE brominated flame retardant CAS 84852-53-9
DBDPE was found to be one of the best flame retardants, i.e. it outperforms other additives in the same class.
What does DBDPE Do?
Decabromodiphenyl ethane is an additive for furniture foam, wire / cable, plastics and many more applications. It is an inert molecule that only becomes active under the high temperatures of a fire. It can prevent a spark from igniting the plastic material; or, if a strong ignition source is held against the plastic, forcing ignition, the DBDPE can dramatically slow the spread of the fire, giving people more time to escape. It also gives firefighters time to arrive and extinguish the fire–protecting occupants and reducing property damage. DBDPE is one of the most popular choices because it performs very well and is so safe. Regulatory agencies around the world have looked at the data and agreed that BDBPE is safe. So, why the proposed ban?
What is the Basis for the Proposed Ban of DBDPE in Canada?
Degradation of DBDPE in Ultraviolet Light
“DBDPE testing under longer-term (e.g., greater than 6 month), environmentally relevant conditions to determine the degradation pathways and transformation products is lacking (possibly influenced by analytical challenges). Nevertheless, potential DBDPE transformation products were evaluated on the basis of predictions from photodegradation studies and biodegradation modeling, and by considering analogue decaBDE transformation products.”
Screening Assessment Certain Organic Flame Retardants Substance Grouping Benzene, 1,1’-(1,2-ethanediyl)bis [2,3,4,5,6- pentabromo-Decabromodiphenyl ethane (DBDPE
“Currently (Q)SARs are not suitable for complex toxicological properties, as they are not fit for purpose for classification and labelling or risk assessment.”
“The current photolytic degradation experiments were performed on DBDPE in solvents and on silica gel. Large differences in the photolytic degradation behaviors of DBDPE may exist between lab matrixes and the natural environmental media such as atmospheric particles, soil, and water as suggested previously for deca-BDEs (Raff and Hites, 2007). Additional evidence is therefore needed to understand the potential photolytic degradation kinetics and to examine the occurrence of degradation intermediates of DBDPE in the real environment.”
Wang et al., Photolytic degradation of decabromodiphenyl ethane (DBDPE), Chemosphere, 89, 844-849, 2012
“The resistance of DeBDethane against debromination compared to DecaBDE might be attributable to differences in molecular structure, i.e. the ether bond in BDE 209 and the aliphatic C-C bridge in DeBDethane.”
“Photodebromination of technical decabromodiphenyl ether (DecaBDE) incorporated into high-impact polystyrene (HIPS) and TV casings was compared under natural sunlight conditions with that of technical decabromodiphenyl ethane (DeBDethane). BDE 209 in pulverized HIPS+DecaBDE samples degraded with a half-life of 51 days. In contrast, no marked loss of DeBDethane occurred throughout the experimental period of 224 days. During BDE 209 photolysis in HIPS+DecaBDE samples, partly debromination to nona- and octa-BDE was observed, however, environmentally relevant polybrominated diphenyl ether (PBDE) congeners such as BDE 47, 99, and 100 were not formed.”
Kajiwara et al., Photolysis Studies of Technical Decabromodiphenyl Ether (DecaBDE) and Ethane (DeBDethane) in Plastics under Natural Sunlight, Environmental Science & Technology, 42, 4404-4409, 2008
This is concrete evidence that banning DBDPE is unfounded. The proposed ban is based on an incorrect guess that it would degrade under UV light to form toxic substances, when in fact we know that does not happen.
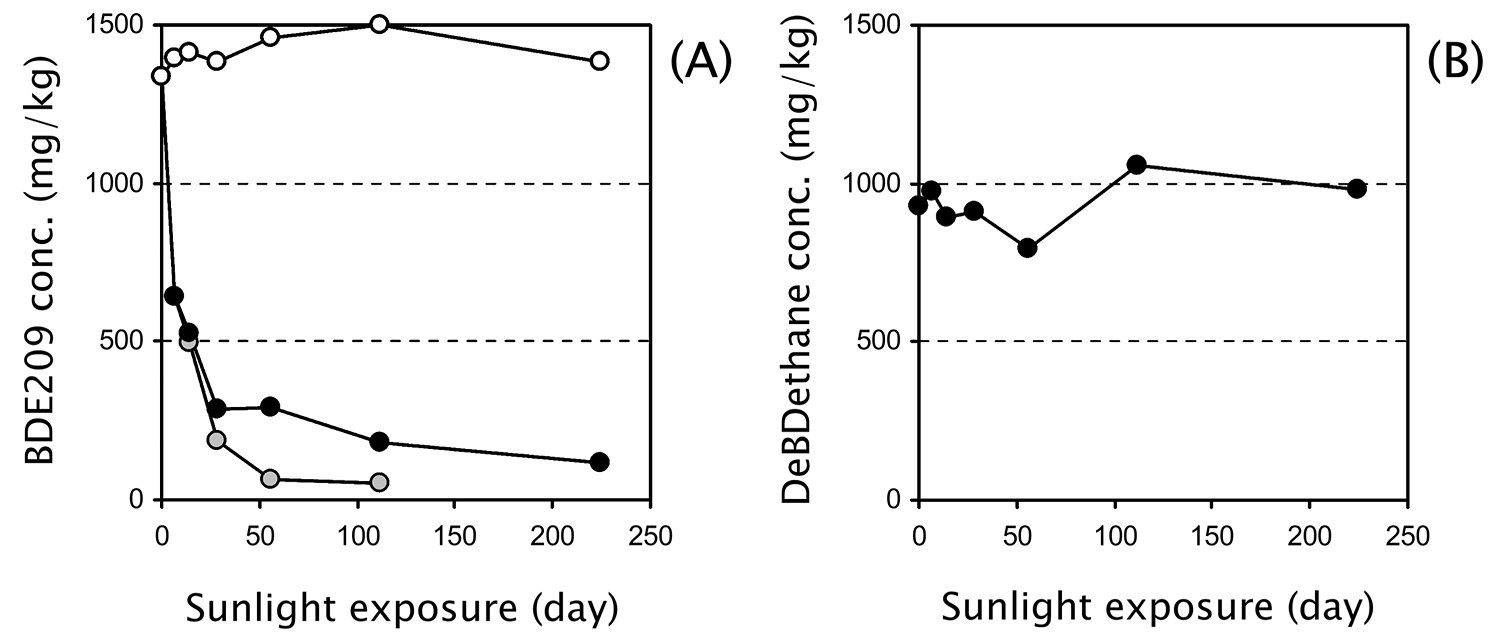
On the left, the scientists show the rapid degradation of the banned DecaBDE under UV light, whereas on the right they show that DBDPE does not degrade. The proposed ban was based on the statement that no experimental evidence existed on this matter; and so, they thought it appropriate to take a guess at what might happen. Apparently they did properly consider this peer-reviewed evidence. There was no need to guess about the degradation of DBDPE, which means that the foundation for the proposed ban has been invalidated.
There is further work proving that DBDPE does not degrade under real conditions and does not degrade to the toxic substances proposed by the ECCC in their modeling / guesswork.
“No photodegradation of EBP from Br9 to Br8, or further to lower brominated congeners of concern, was evident in HIPS.”
“No photolytic debromination and no substitution of bromines with oxygen were seen in PP compounds…”
“A read-across comparison of the photolytic behavior from DecaBDE to EBP would be misleading in risk assessment exercises.”
I understand that this data has been submitted for consideration, but has been ignored so far. This is categorical proof that the “modeling” used by the ECCC was completely unreliable and should be discarded, because actual experimental data disproves it. DBDPE does not degrade, and even when forced to degrade, it does not form the toxic substances imagined by the ECCC modeling. Even in the light of this overwhelming scientific evidence, there has been no change in the position of the government. This is their response to companies who have objected and sent evidence:
“not raising sufficient uncertainty or doubt in the scientific considerations of the underlying the proposed regulations…”
This response is not appropriate. The Minister is saying that he would prefer to believe the disproven guess of a computer program over real, peer-reviewed scientific evidence to the contrary. That is unwise, unscientific, and certaqinly not in the best interests of the public.
Let us compare what the Minister has said to the analogy above of the weather forecast. In our analogy, the Minister is standing outside in the pouring rain (real-world data) and insisting that it is not raining because his weather app (computer model guess) says that it is not raining. Believing a computer’s guess when the real world evidence says the opposite is not the act of a wise person.
Other Considerations around the Proposed Ban of DBDPE
Conclusions – Is the Proposed Canadian Ban of DBDPE Justified?
Brominated flame retardants including DBDPE are proven to save lives. Alternative flame retardants do not perform as well, and, in many cases, they fail to pass the flame retardancy safety tests, putting lives at risk.
There is no scientific evidence that DBDPE is toxic to organisms, animals or humans at levels found in the environment. That is partly because it is a next generation flame retardant which was designed to be safe. It has high molecular weight to prevent migration, and crucially, it is extremely insoluble, which prevents exposure.
The proposed ban is based on pure guesswork. Based on the appearance of the DBDPE molecule, someone has speculated that it will degrade to form toxic substances. However, there is no scientific experimental evidence to support that speculation. Therefore, banning DBDPE would be as foolhardy and unjust as convicting an innocent person based on hearsay. Sound science and justice both depend upon evidence and in this instance, there is no evidence of harm.
Furthermore, the proposed ban was based on an assertion that no scientific evidence existed to show how DBDPE actually degrades. That assertion was incorrect. Peer-reviewed science does exist and was overlooked. The data clearly shows that DBDPE does not degrade under environmental conditions, thus the premise of the proposed ban is invalid.
In summary, the ECCC proposed a ban based on these three invalid assumptions:
- That DBDPE degrades rapidly like DecaBDE – disproven by experiment
- That DBDPE forms toxic substances like DecaBDE – disproven by experiment
- That a model /guess should be used because experimental data was not available – disproven – models cannot substitute for real data and experimental data does exist
As an independent expert, Phantom Plastics wrote to ask the Minister to appoint a Board of Review to “inquire into the nature and extent of danger” posed by DBDPE because, at present, there is no scientific justification for a ban. If you want to prevent this injustice, then please contact the Minister as well.
Once again, we see that the demonization of plastics and their additives is unwarranted and distracts the public from the real issues. Want to know more? Then read the The Plastics Paradox book or watch the The Great Plastics Distraction video.
“you can be for the environment, or against plastics, but not both”.

Dr. Chris DeArmitt is a renowned independent scientist and keynote speaker with decades of experience solving tough technical challenges for some of the world’s leading companies. He is a Fellow of the Royal Society of Chemistry and Fellow of the Institute of Materials, Minerals and Mining. He is also a Chartered Chemist with a long list of publications, presentations and patents to his name. His review of the science around plastics and the environment was performed unpaid to preserve impartiality.
Income 2024 : ~ 60% NAICS 424690 Chemicals, ~30% NAICS 423990 Durable Goods, 10% NAICS 541110 Legal, 0% NAICS 325211 Plastics Composition varies over time



