Magnetite Black Iron Oxide Properties and Applications
Introduction to Magnetite
Magnetite is a natural mineral with many special properties which make it useful for a wide range of applications. This article describes all of the properties of magnetite and how each property leads to its use as a specialty mineral functional filler in commercial products. By understanding the properties we can understand why magnetite is used now and also how it might be used in new applications in the future.
What makes me an expert on magnetite? I worked for one of the world’s largest magnetite producers, LKAB, based in Sweden. They sell billions of dollars of magnetite every year. During that time I collected and read hundreds of peer-reviewed articles about magnetite and its uses. That eventually led me to write an encyclopedia chapter on this special mineral. This page is a more accessible way for you to learn everything about magnetite without having to buy an encyclopedia or read hundreds of articles.
What is Magnetite?
Magnetite is a naturally occurring black iron oxide with the chemical structure Fe3O4. It is one of several forms of iron oxide. Fe3O4 is known as iron (III) oxide, whereas Fe2O3 is iron (II) oxide, commonly referred to as hematite.
Iron is the fourth most abundant element in the Earth’s crust and over 90% of all metal we mine is iron, specifically hematite and magnetite, which is alloyed with carbon to make steel. This article looks at the more specialty and perhaps interesting uses of magnetite in plastics, paints, concrete and more.

Chemical formula: Fe3O4
CAS Number: 1309-38-2 or 1317-61-9
RMM: 231.53
Density: 5.2 gcm-3
Mohs hardness: 5.5 – 6.5
Colour: Black, shiny
Refractive index: 2.42
Melting point: 1600°C / 2900°F
CTE: 9 x 10-6 K-1
Thermal conductivity: 5.1 Wm-1K1
Specific heat capacity: 0.73 kJkg-1K-1 3.8 kJL-1K-1
Dielectric constant: 3 – 14 (depends on frequency)
Solubility: Insoluble in water, bases & solvents, dissolves in acid
Reactivity: Unreactive (except to strong acids)
What are the Magnetic Properties of Magnetite?
The very name “magnetite” implies that the mineral must have magnetic properties and that is indeed the case. Of all naturally occurring minerals, magnetite is most strongly attracted to a magnet. This property is very useful. For example, magnetite ore is easily separated from other contaminant minerals simply by applying a magnetic field. Once the field is removed, the magnetite is no longer attracted. This ability to be attracted to a magnet and then lose all magnetic attraction once the field is removed is described by the term remanence. Because the magnetism remaining is insignificant, magnetite is called a low remanence material or a soft magnetic material. In contrast, hematite is very weakly attracted to a magnet (about 100x less than magnetite is) but hematite is a hard magnetic material and retains more magnetism once the magnet is removed.
People are often confused about the magnetic properties of magnetite. So, to clarify the confusion, magnetic is not itself magnetic. Magnetite will not attract a paperclip or piece of iron for example. However, magnetite is “magnetic” in the sense that it is readily attracted to a magnet. In technical terms, magnetite is described as ferrimagnetic. There is a temperature above which magnetite is no longer attracted to a magnet and that so-called Curie Temperature is about 585°C.
What is Lodestone and How is it Different to Magnetite?
Although the vast majority of natural magnetite does not behave as a magnet, there is a special and rare type of magnetized magnetite that can be found in nature. This special form of magnetite is called Lodestone and it was how humans first discovered the concept of magnets in the 6th Century BC. The first compass was made by suspending a Lodestone, allowing it to rotate thus indicating magnetic north and south.
So, how did Lodestone become magnetized when the vast majority of magnetite did not? That was a mystery for hundreds of years and was solved only recently. It turns out that lightning magnetizes the magnetite in the vicinity of the Earth’s surface where the strike occurs.
Are There Applications Exploiting the Magnetic Properties of Magnetite?
Although Lodestone was critical for our discovery and understanding of magnetism, it has been superseded by much better, stronger man-made magnets such as samarium cobalt and neodymium iron boride types. One newer and still relevant application for magnetite is in ferrofluids. Indeed, the first ferrofluid was created when a scientist named Stephen Papell at NASA ball-milled magnetite into very fine particles and suspended them in an oil together with surfactant to aid with dispersion of the particles.
Ferrofluids and magnetorheological fluids
Ferrofluid has several applications. Probably the most well-known is in audio transducers such as loudspeakers and microphones where the fluid is placed in the gap between the permanent magnet and the voice coil where it serves several functions. One function is to center the voice coil and allows the spider to be eliminated in some instances. By adjusting the rheological properties of the ferrofluid, the resonant frequency of the speaker can be damped and controlled. Also, the thermal conductivity of the particles helps dissipate heat.
Magnetorheological fluids are related but employ larger particles that are too massive to be colloidally stable. One use for them is active dampers to provide a smooth ride for luxury cars and to avoid truck driver discomfort. More on MR, also known as MRF fluids and how they work can be found here. Seismic dampers made from MRF fluids can be placed in the foundations of buildings and activated to prevent resonances induced by earthquakes from destroying buildings.

Novelty uses for Magnetite
Magnetite is used as a filler for paints that allow placement and arrangements of magnets on a surface in much the same way as one uses magnets on a refrigerator door. Rather than the need for a sheet of steel, as is the case with a fridge door, by using magnetite as a magnetic paint additive, it is possible to have the same effect on any surface such as a wall in your home or a class-room blackboard. Magnetite paints are available commercially and can be found in popular home improvement stores.

A fun novelty item is an hourglass filled with particles that are attracted to a magnet. Either iron filings or magnetite can be used. As the particles fall down, they are attracted to a powerful neodymium magnet and form spiky patterns reminiscent of those seen in ferrofluids as in the picture above. Small versions are inexpensive and readily available but the renowned designer Wayne Strattman of Strattman Designs made a larger custom version using magnetite I provided.
It is possible to perform a magic trick with these hourglasses. When a strong magnet is brought near to the stream of downward flowing particles, it causes them to attract each other which blocks the hole and stops flow. When the magnet is removed the flow restarts. In effect, one can create a valve without mechanical parts only by the application of a magnetic field. The magic effect is best performed using a strong neodymium magnet either concealed inside the hand, or worn in plain sight in the form of a ring on the magician’s finger. These rings are also inexpensive and available online. A Curie-effect heat engine or Curie pendulum is another toy or magic trick that can be performed using magnetite.
Magnetite for Detection and Separation of Plastics
As well as the entertainment uses for magnetite, there are other more commerial examples. One is the addition of magnetite to plastics to make them detectable with a metal detector. This is especially valuable in food production facilities where they want to be able to detect any contamination in the food and reject the item before it leaves the factory. For example, if a piece of plastic breaks off from the machinery and gets trapped in a biscuit or loaf of bread they cannot detect the plastic unless magnetite is added.
Another recent development has been the addition of magnetite to facilitate in the sorting of plastics for recylcing. Many plastics look the same and that prevents them from being easily sorted. Simply by adding a small weight percentage of magnetite, that plastic is now easily removed from a pile of other plastics using an electromagnet. The ability to sort plastics well is vital for effective recylcing because each plastic must be recycled seperately without any contamination from other plastics.
Electrical Properties of Magnetite in Plastics and Coatings
Magnetite is a semi-conductor of electricity and reported values have a wide range because the conductivity of semiconductors depends on trace impurities. So, magnetite coming from different mines will be expected to have significantly different electrical conductivity. Extremely pure magnetite has an electrical resistivity around 1.0 x 104 micro ohm cm. The conductivity of single crystals is 250 per Ohm per cm. When added to plastics and coatings, magnetite does not give any measurable electrical conductivity until high loadings are reached and the magnetite particles touch each other creating a electrical path or circuit. This effect is known as the percolation threshold and depends on particle size, dispersion and other parameters.
A published study on natural magnetite in PP and PA6 – Magnetite Functional Filler: A Compounding Study in Polypropylene and Polyamide showed that electrical conductivity began at 33 volume percent magnetite and increase up to the maximum possible loading of 47 volume percent (85 weight percent) where an electrical resistivity of 10 kOhm m was measured. So, one can choose not conductive or conductive depending on the filler loading.
What is the Density of Magnetite?
The density of magnetite is 5.2 gcm-3 and, as we shall see, that leads to a number of applications for this specialty functional filler. While some effects of high density may be obvious, some are surprising.
How does adding magnetite affect the density of plastics?
Most organic materials like plastics, coatings, wood and so on have a density around 1 gcm-3, which also the density of water. Many common mineral used as fillers for plastics have densities about 2.5 or 3 times higher than that of plastics. PE and PP have densities around 0.9 gcm-3 whereas calcium carbonate, kaolin clay and talc have densities of approximately 2.7 gcm-3. It is therefor possible to increase the density of plastics somewhat through addition of those standard mineral fillers.
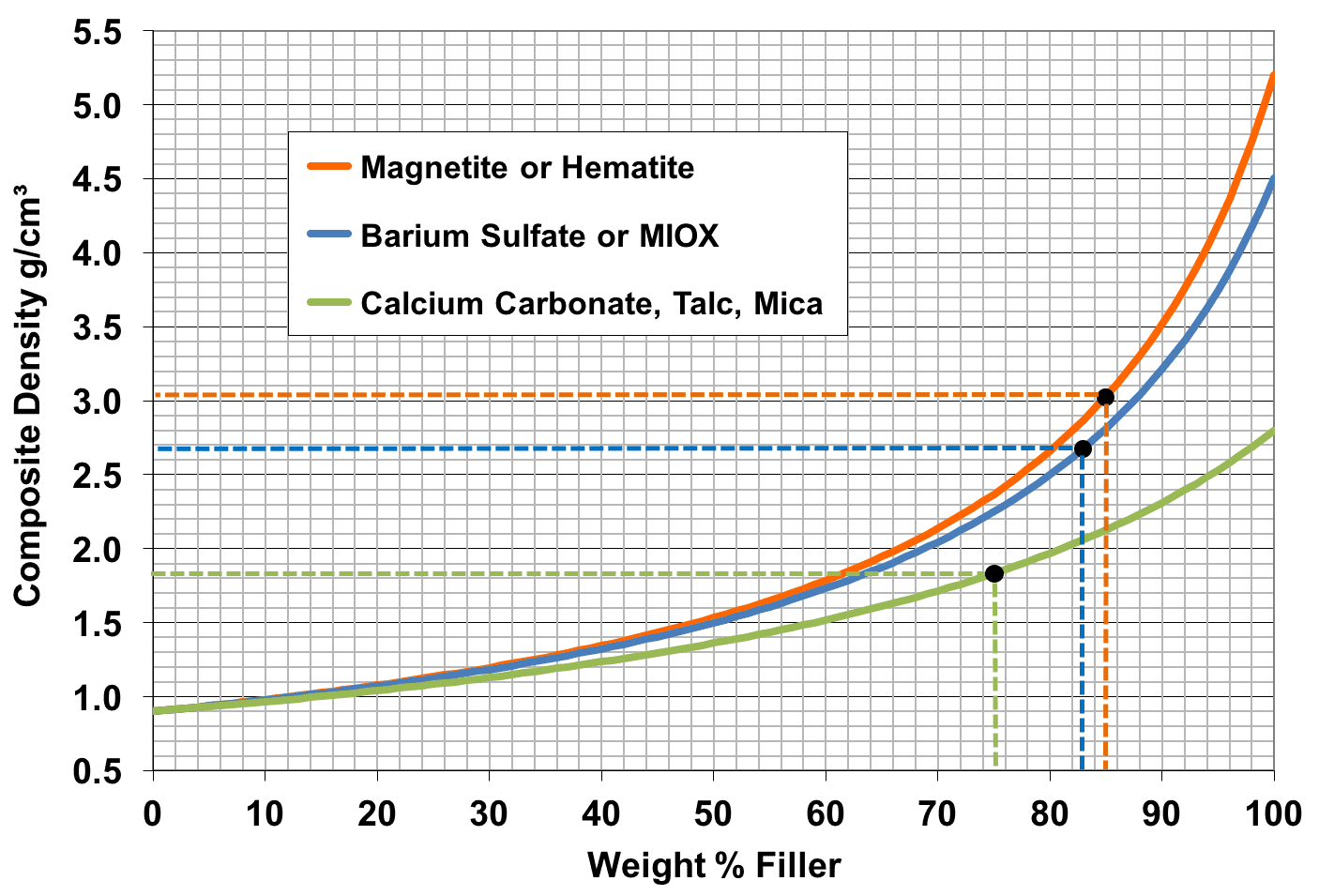
As shown in the diagram, the maximum composite density achievable using typical fillers is about 2 gcm-3. However, some applications such as sound deadening require higher density. There, the standard high density filler used is barium sulfate. The barium sulfate itself has a density of about 4.5 gcm-3 and but magnetite performs even better due to its exceptionally high density of 5.2 gcm-3. For the same volume % loading of fillers, magnetite gives a density of ~3 gcm-3 compared to about ~2.5 gcm-3 for barium sulfate and ~2 gcm-3 for other fillers. The increase in density is not linear, so this link goes to a composite density calculator so that you can hit your target density first time without trial and error.
What other ways does adding high density magnetite affect properties of plastics?
As shown in the diagram, the maximum composite density achievable using typical fillers is about 2 gcm-3. However, some applications such as sound deadening require higher density. There, the standard high density filler used is barium sulfate. The barium sulfate itself has a density of about 4.5 gcm-3 and but magnetite performs even better due to its exceptionally high density of 5.2 gcm-3. For the same volume % loading of fillers, magnetite gives a density of ~3 gcm-3 compared to about ~2.5 gcm-3 for barium sulfate and ~2 gcm-3 for other fillers. The increase in density is not linear, so this link goes to a composite density calculator so that you can hit your target density first time without trial and error.
What applications are there for high density magnetite-filled plastics?
The main application for magnetite-filled plastics is for sound damping. For example, magnetite-filled polypropylene is used in luxury cars to dampen and tune the sound of the engine and cabin. This application has existed for over a decade. In the cabin, the magnetite is applied in a polyurethane foam. In the engine compartment magnetite is used in combination with glass-fiber reinforcement with PP as the plastic matrix. The glass fiber provides reinforcement and the magnetite is there for density. Magnetite is also used in luxury perfume bottle caps where it serves to give heft and the perception of high quality as well as the magnetic closure function. Frisbee golf or disc golf requires discs of varying weights and magnetite can be used to create various discs of the same size, made in the same molds. In other applications, a plastic device must have negative buoyancy, i.e. it must sink in water – so magnetite is added to provide that function. As well as plastics, magnetite is used as a filler in concrete where it allows massive, highly stable foundations to be made.
Peer-reviewed articles illustrate magnetite loaded PP and nylon plastics with good properties despite the high loading of magnetite filler. Scientists studying those materials were surprised to find that magnetite does not affect the thermal stability of the plastics. Often, polyolefins like PE and PP are degraded by transition metals like copper and iron which can act as catalysts for oxidation of the polymer. Even parts per million levels can be enough metal to influence stability. Despite addition of a high loading of magnetite, which contains over 70 percent iron by weight, the thermal stability of the plastics was hardly affected. Magnetite works well in a range of plastics including PE, PP, PA, EVA and more. However, it may not be wise to compound it into PVC and other halogenated resins (PTFE, FEP, PVDF, CPVC etc.) as a chemical reaction could give off toxic fumes. See for example, the article entitled Catalytic de-chlorination of products from PVC degradation by magnetite (Fe3O4).
Magnetite Absorbs Microwaves and Radar
Microwaves cover the approximate wavelength range one meter to one millimeter corresponding to frequencies between 300 MHz and 300 GHz. When frequencies in that range are used for location purposes, then the term radar is applied. GSM cell phone communications are also in that frequency range.
Those frequencies pass right through plastic materials unhindered because plastics have neither electrical conductivity nor magnetic properties. As we have noted, magnetite has electrical and magnetic properties, so we might expect it to interact very strongly with microwaves – a form of electromagnetic radiation.
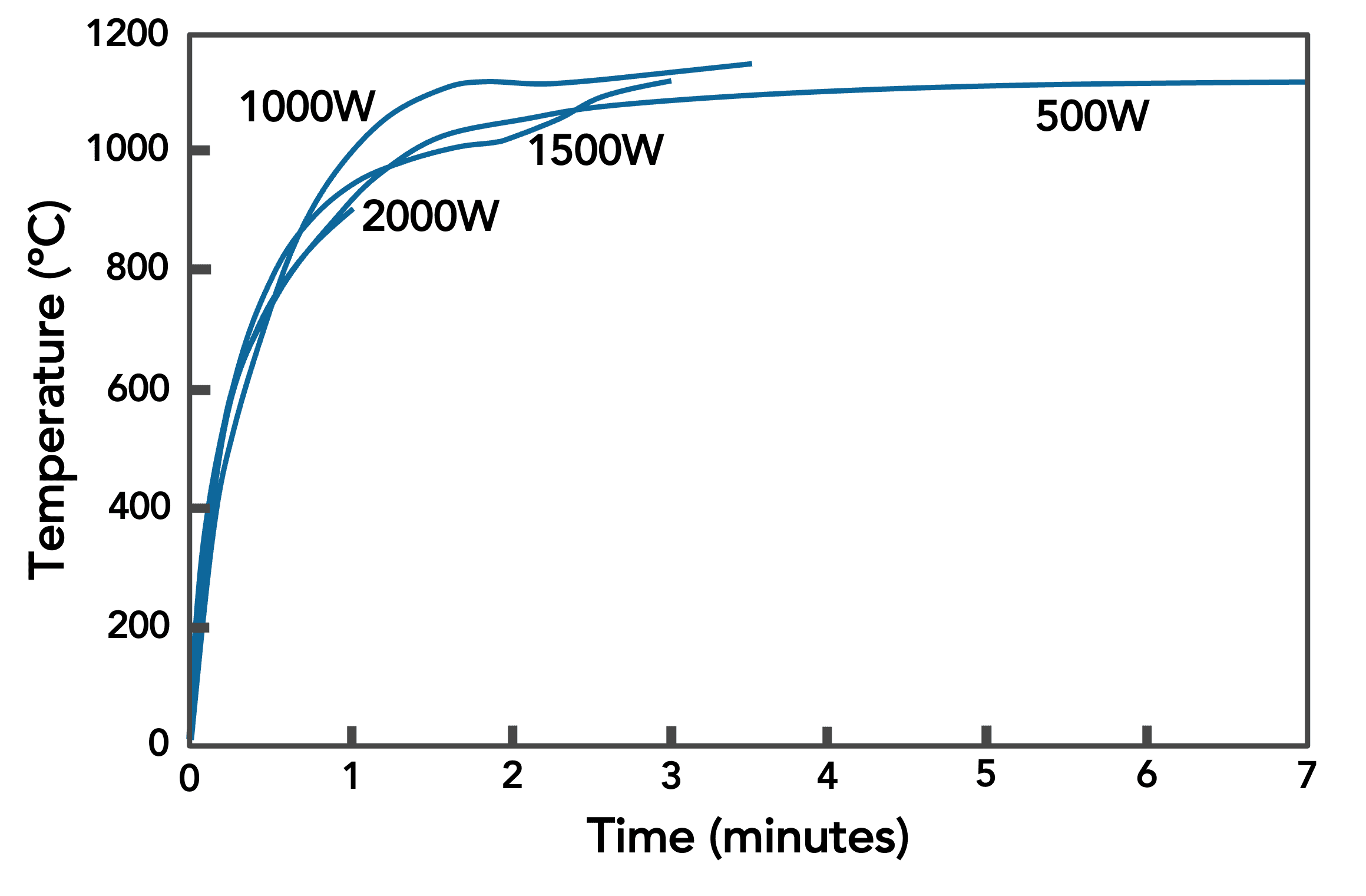
Measurement of absorption requires specialized equipment but it is possible to detect the microwave absorption property of magnetite even at home. I do not recommend this to anyone but when a magnetite sample is placed in a normal domestic microwave oven, it can reach temperatures of several hundreds degrees Celsius in a minute or two. People have even made microwave kilns that can melt silver and other metals by packing magnetite powder around a ceramic or graphite crucible and insulating the apparatus to keep the heat trapped inside.
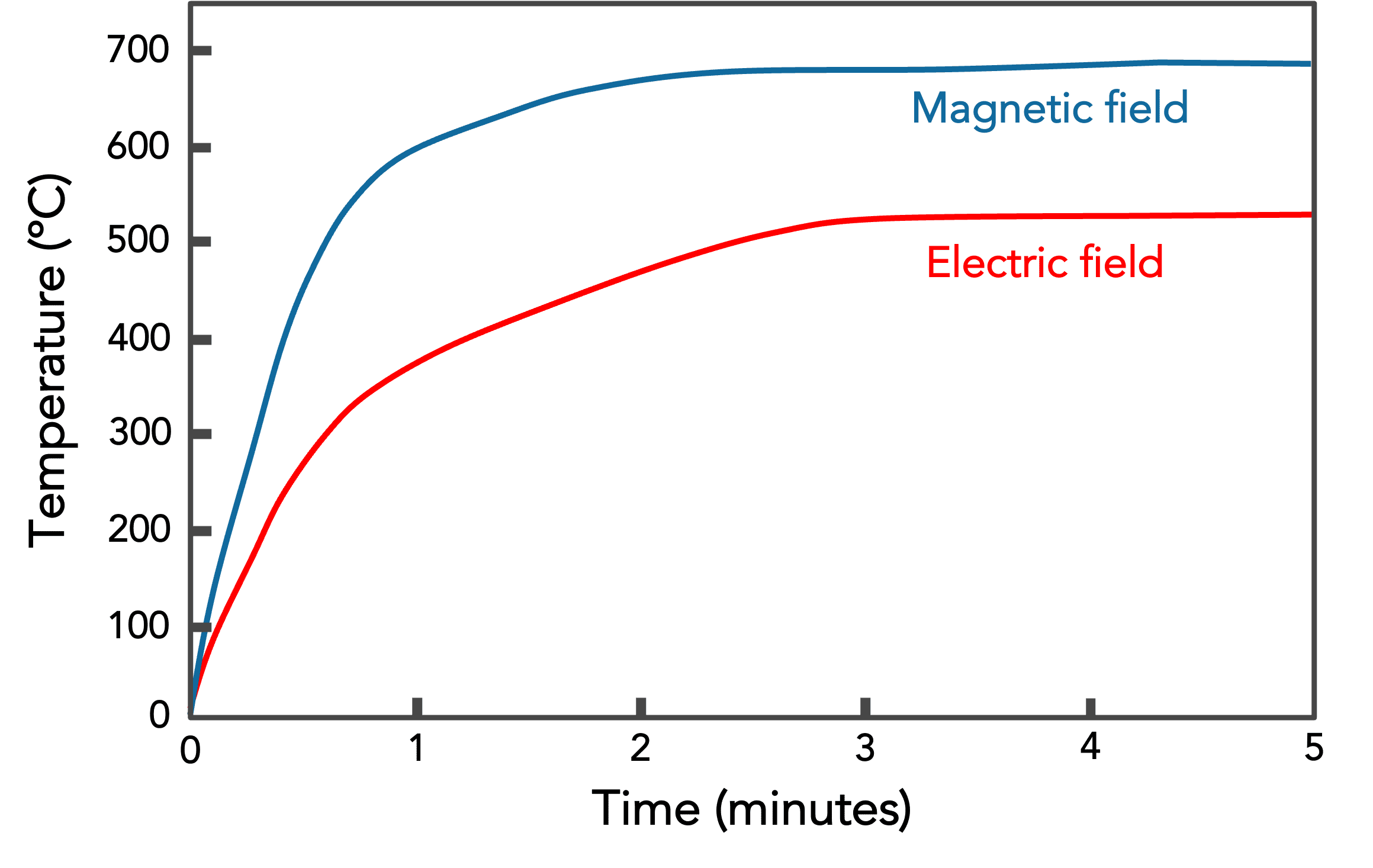
In a clever experiment, scientists were able to heat magnetite using only the electric component of the microwave energy or only the magnetic component of the microwave. This resulted in the heating curve shown. As expected, magnetite is unusual in that it heats rapidly via both mechanisms. Electrically conductive materials like metals reflect microwaves and do not heat because the energy is not absorbed. Electrically insulating materials like plastics allow microwaves to pass through without any interaction or heating.
The graph on the right shows how fact magnetite heats up in a microwave oven. An astonishing temperature of ~1000°C (~2000°F) can be reached in just a minute or two. In contrast to the graph on the left where only the magnetic or electric component of the microwave energy is used, the graph on the right shows the normal case e.g. in a domestic microwave oven when both components act at the same time.
Addition of magnetite to plastics as a microwave and radar blocking filler also provides the ability to rapidly heat the plastic. This latter effect was patented for cooking trays that allow food to brown when cooked in a microwave, which is normally not possible. US4542271A Microwave browning wares and method for the manufacture thereof. This works because addition of magnetite makes the surface of the plastic become so hot that it is able to brown meat while cooking.
Magnetite-filled polymers have been used for shielding boat masts. A metal boat mast will create a “ping” every time the ship’s radar sweeps past. The ping is annoying and not helpful because the position of the mast is known and of little interest to the captain. By coating the mast with magnetite-filled material, the radar is blocked i.e. absorbed rather than reflected, so the pinging is eliminated. A related application is magnetite-filled plastic sheets used to prevent reflections in radomes. The radar signal can reflect off the walls and cause ghost signals, so magnetite radar shielding materials are used in strategic locations to absorb stray signals, improving S/N ratio and sensitivity.
Magnetite Black Iron Oxide Pigment
Black iron oxide pigment is Fe3O4 i.e. magnetite. When very fine or nanoparticles are desired then precipitation is used to create them. For coarser particles, natural magnetite mineral is preferred. All sizes are available. When added to plastics and coatings, it is found that magnetite is a relatively weak pigment and that the tinting strength depends on particle size. When higher blackness is desired, then finer particles are used. Conversely, sometimes one wishes to add magnetite but not have the final product appear black. For example, food industry zip ties are made bright blue to make them visible in case they get into the food by accident, they are easily seen and removed. Magnetite can be added to the nylon to make it detectable by metal detector or x-ray but the zip tie must still be bright blue. By selecting a coarser grade of magnetite with low pigment strength, it is possible for the zip ties to contain the black magnetite and yet still maintain the correct appearance.
Another instance where magnetite is useful as a black pigment is in ceramic glazes. The most common black pigment of all is carbon black. However, carbon black and other organic pigments are too thermally unstable to be used in ceramics because they are fired at 800-1100°C. A designer wanted to put large black pigment particles in a ceramic glaze and all other attempts had failed due to pigment decomposition or discoloration. Coarse magnetite pigment particles performed perfectly, as shown in the image because magnetite does not melt until over 1500°C is reached. Adhesion between magnetite ceramic pigment and the glaze was also excellent.

Thermal Properties of Magnetite Iron Oxide
The thermal conductivity of magnetite is 5.1 W m-1K-1 which is higher than most other common minerals such as calcium carbonate, dolomite, mica or kaolin clay and much higher than plastics 0.2-0.4 W m-1K-1. This means that when added to a plastic compound, magnetite helps with conduction of heat. For example, when molding parts, the cycle time is reduced because heating and cooling are both accelerated.
The most remarkable thermal property of magnetite is its specific heat capacity which is the parameter reflecting the amount of heat required to heat up a substance by a set amount. Specific heat capacity is a misunderstood topic and even some books by experts fail to present it properly. There are two ways to express the specific heat capacity of a solid. The most common way is to give the amount of heat in Watts needed to raise the temperature of one kilogram of solid by one Kelvin (or one degree Celsius, which is numerically the same). This is known as the mass specific heat capacity and values for solids vary widely.
The other, less common way to express the heat capacity is the volumetric heat capacity meaning the amount of energy in Watts required to raise the temperature of one liter of solid by one Kelvin. It turns out that the volumetric heat capacity of almost every solid is the same, around 2 kJ L-1K-1. People often believe that adding mineral fillers to plastics will change their specific heat capacity but on a volume basis, that is incorrect. If one wishes to increase the amount of heat a piece of plastic can hold, then the best way is to add magnetite because it is one of the only solids with a high volumetric heat capacity. In fact, the volumetric heat capacity of magnetite is 3.8 kJ L-1K-1, which is almost 2x higher than that of other solids (see table).
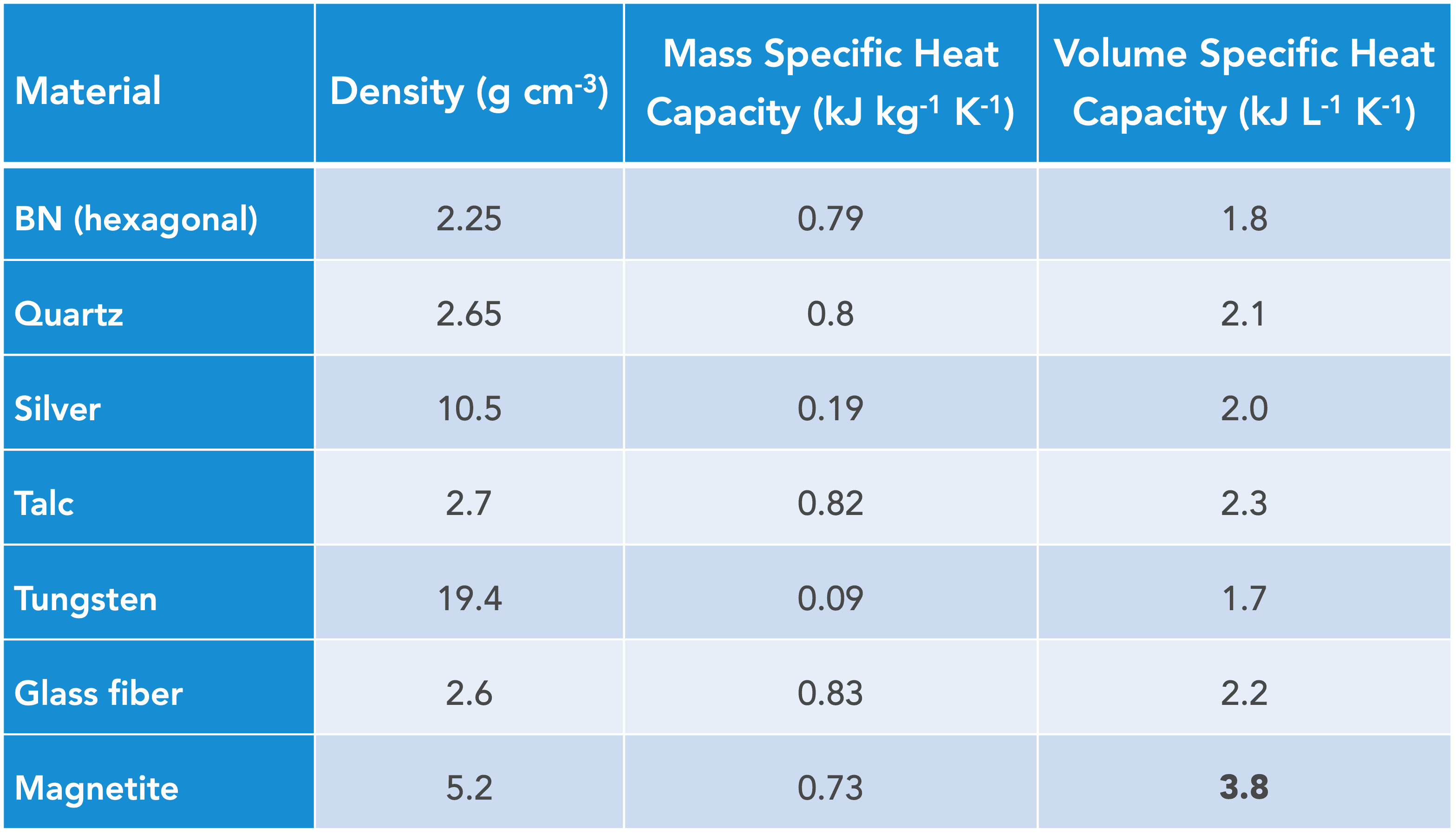
Applications for the high volumetric specific heat capacity of magnetite
In recent years architects have focused on greener buildings. They want to make buildings that hold heat to save electricity from heating and conversely, they want buildings that stay cool to avoid using so much air-conditioning. I have seen articles claiming that using concrete will help but they are incorrect. A given volume of concrete holds the same amount of heat as other solids.
The solution is to add high amounts of magnetite as a filler for concrete. The resultant concrete has better thermal properties, excellent mechanical properties and increased density too. Magnetite-filled concrete has been used in radiology wards for many years because its high density blocks x-rays, so it can replace toxic lead shielding. Magnetite materials are also used to make storage heaters more effective. It should not be surprising that magnetite is also added to plastics when the formulator wants to increase the specific heat capacity of plastic materials.
Safety of Magnetite
All naturally occurring minerals are included in the TSCA list by definition even though they may not be actually on the printed list. Magnetite is used as a food supplement and is considered safe. It is generally recognized as safe (GRAS). Magnetite is approved for indirect food contact when used as a filler in plastics, in fact I was the person who applied for and received an FDA opinion letter. While magnetite itself is safe, like any naturally occurring mined material, it can contain impurities, so that should be considered when making a purchase.
Conclusion – Magnetite is a Multi-functional Filler for Plastics
Magnetite is a black iron oxide mineral with unusual properties and diverse applications stemming from those properties. Often used for its high density (5.2 gcm-3), it also has high thermal conductivity and quite high electrical conductivity. It is x-ray opaque and can be used to block radiation including microwaves and radar. Magnetite is isotropic and non-reinforcing due to the low aspect ratio of the particles. Further details can be found in this encyclopedia chapter I authored as well as R. M. Cornell and U. Schwertmann, The iron oxides: structure, properties, reactions, occurrences and uses.
Want to buy magnetite filler? Contact Phantom Plastics to select the best grade for your application and get contact to our recommended supplier.
