Polyhedral Oligomeric Silsesquioxanes
Introduction to POSS
Polyhedral oligomeric silsesquioxanes, often abbreviated POSS®, PSS or POS, are hydrid materials composed of an inner, rigid, silica cage structure surrounded by more flexible organic groups. The organic groups can be selected to impart solubility of the POSS molecule and to tune the properties.
Visually, the POSS molecule looks like filler particles and many consider it to be a kind of molecular filler or the smallest possible silica particle.
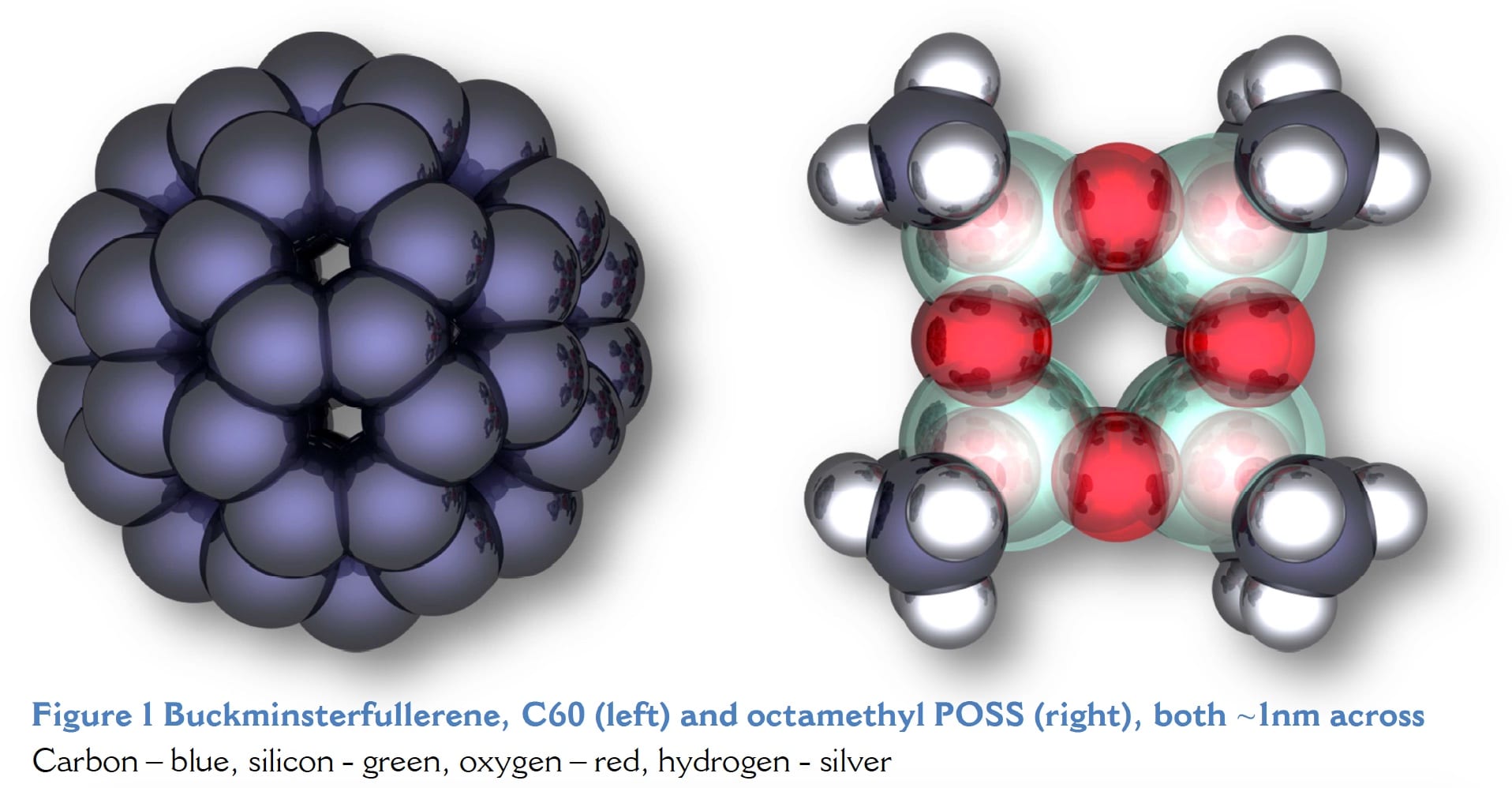
Here is a 3D ray traced video animation I made of octaiso-butyl POSS using POV-Ray. It took an incredible amount of work to create, but I think the results speak for themselves. Click to play a movie of iso-butyl POSS rotating. The glass-like spheres represent silicon, red is for oxygen, blue is for carbon and silver spheres are hydrogen atoms.
Polyhedral oligomeric silsesquioxanes, now commonly referred to under the trade name POSS, were discovered in the 1940s. Although they were not pursued commercially at that time, there was a revival of interest in the 1990s and that interest has been sustained globally ever since. The cage-like shape of the molecule is visually intriguing and that is certainly part of the appeal. Conventional molecules tend to be more or less linear with perhaps some degree of branching and the behavior of those “normal” molecules is rather well understood. Molecules departing from that norm tend to attract substantial interest in the hope that they will provide revolutionary properties and technological breakthroughs. For example, hyperbranched polymers, and their structurally perfect analogues the dendrimers, have been the focus of intense research and their unconventional structures do indeed lead to unusual properties. Cage structured hydrocarbons are well-known and again have unusual properties. Cubane is one example, having a density higher than any other hydrocarbon (1.29 gcm-3). Similarly, some POSS types display exceptionally high densities of up to 1.82 gcm-3 (see Table 1). Another family of cage-like hydrocarbons, the diamondoids, found naturally in petroleum deposits, was investigated by Chevron Technology Ventures in collaboration with Stanford for several years. The simplest diamondoid is adamantine with several other higher homologues reported and can be chemically derivatized to impart reactive groups. Interestingly, the diamondoids are also referred to as nanodiamonds, thus implying that they are nanoparticles of diamond. In much the same way, POSS is often referred to as “nano silica”. In both cases however, the terminology is misleading. As will be illustrated later, these substances are all molecules that merely appear reminiscent of particles.
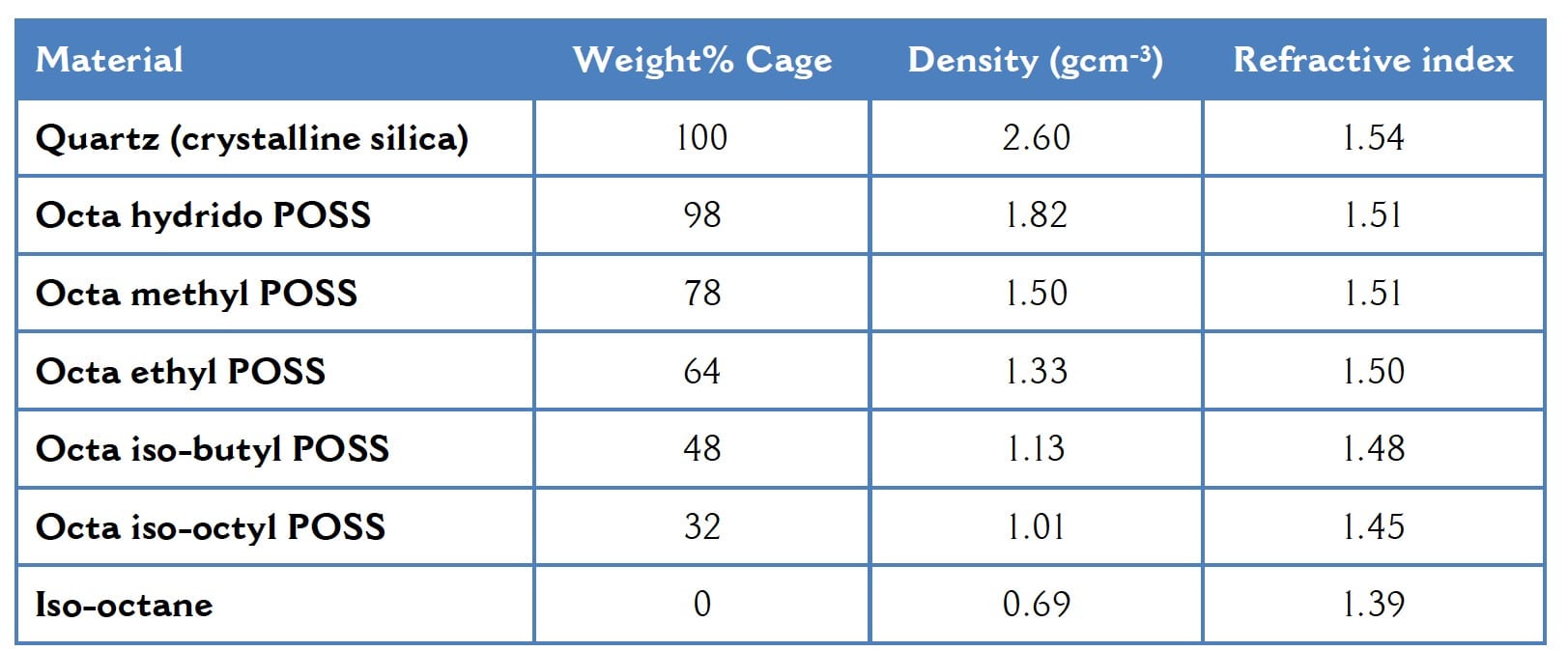
Table 1 How POSS Structure Affects Density and Refractive Index
As well as density and refractive index, other properties including modulus, thermal conductivity, solubility and so on are expected to vary systematically as one varies the ratio of inorganic cage to the surrounding organic groups. Therefore, POSS is able to span the gamut of properties from those associates with inorganic materials, to those found for organic substances.
Conceptually, one may consider each POSS molecule to be a filled polymer where there is a central “silica” filler bonded within an organic matrix. Thus, the properties of this molecular composite are expected to follow the same rules as for macroscopic composites so that for example, the modulus of the POSS would be dependent upon the moduli of the central “silica” cage, the organic phase and the volume fraction of each as described by the Halpin-Tsai equation. Other well established polymer composite equations could also be applied in order to estimate the properties of those POSS that already exist and to predict properties of POSS types not yet extant.
No discussion of cage-like molecules would be complete without mentioning C60, Buckminsterfullerene (see Figure 1) and the other fullerenes. The discovery of these new forms of carbon was greeted with tremendous enthusiasm and much speculation about unique properties leading to new applications. I was studying at Sussex University in that exciting time period and was taught by Professor Sir Harry Kroto and the other members of that research team, Professors Dave Walton and Roger Taylor. Yet despite the momentous discovery, now, over 20 years later, there are no commercial applications.
While all of these cage molecules generated excitement and did indeed turn out to have unique properties, large commercial applications have not emerged for three reasons. It has proven difficult to synthesize larger amounts and to produce the diverse chemistries needed to facilitate adoption in a wide range of applications. Furthermore, the prices of these materials are too high to justify their use. Even though performance advantages may have been seen in some cases, they were simply not enough to make these new materials competitive with those already incumbent in the market.
Therefore, we can conclude that it is not enough that a molecule is intriguing or different. In order to have commercial success it must fulfill one or both of these criteria:
- Provide performance levels unattainable by any other route
- Lower the final cost (holistic, total system cost, not materials cost alone
Furthermore, it is my experience that the best breakthrough technologies on the market today took 15-20 years to come to fruition. When one discovers a new material, it is both a blessing and a curse. Certainly, the material is likely to exhibit new properties but one must first find those properties, then work out how that can bring value and who the potential customers might be. Testing ensues, alliances are made and with luck, eventually a product reaches the market.
Chevron Technology Ventures’ interest in the diamondoids has fizzled out and while academic research continues on those and hyperbranched polymers, dendrimers and of course C60, there is little prospect for commercial applications. Looking at all these materials, it is my view that the POSS family of chemicals is, in fact, the most promising of all. They offer the whole range of chemistries and can be tuned easily to attain a spectrum of properties. Equally important, they can be made on a large scale using conventional chemical reactors, resulting in prices far lower than for those other materials.
The organic side groups on POSS span the whole range of chemistries: fluoroalkyl, alkyl, phenyl, alcohols, thiols, amines, vinyl, carboxylic acid, sulfonic acid, PEG, acrylate, methacrylate, epoxides, halides, imides, silanes and silanols. Interestingly, some organosilanes starting materials convert readily to POSS whereas other POSS types simply cannot be synthesized. As one example octaisooctyl POSS is available in bulk whereas octa n-octyl POSS cannot be made at all. There is at present no understanding as to why some kinds can be made and others cannot.
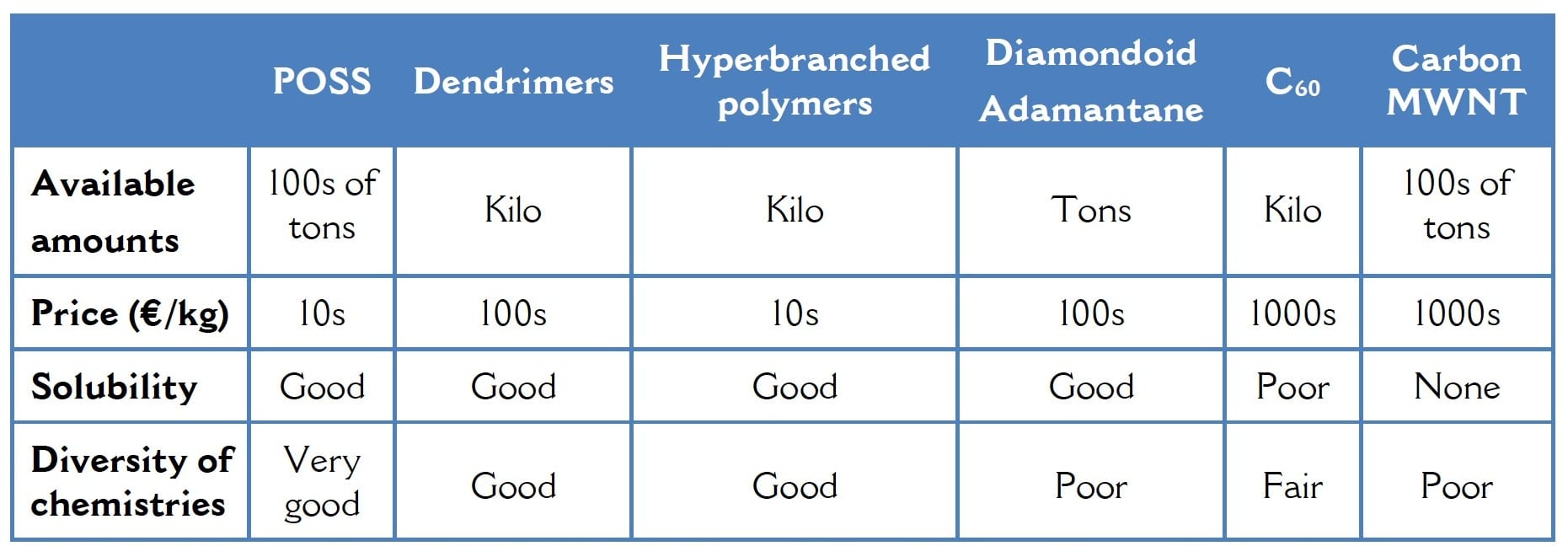
Table 2 How POSS Compares to Other Molecular Fillers Regarding Availability and Price
As a comparison, a starburst dendrimer with 8 functional (amino) groups per molecule costs $181 for 2.0g of a solution in methanol where the concentration of dendrimer is just 20 weight %. Thus, the effective price for the dendrimer is ~$450/g. Contrast that to an octafunctional (vinyl) POSS, also a spherical molecule of well-defined structure with pricing of $2/g, POSS octaamic acid for $24/g or the acrylates and epoxy POSS at only 60 cents per gram. Not to mention that those are the R&D, small order prices for the POSS which drop very significantly for larger orders.
Probably due to the diversity and relatively low POSS prices, some commercial POSS applications do exist and they continue to grow slowly as is the way with such new technologies. However, companies are often hesitant to use any technology that has only one supplier because it can lead to unfavorably high pricing and instability of supply. It could well be that such considerations have slowed the application of POSS.
Having set the scene in terms of what POSS is and how it fits into the landscape of other high profile molecules, we will now look in more detail at POSS types and how they can be used to modify the properties of polymers. Normally, scientific reviews simply look at what can be achieved. As stated in the preface, the aim of the POSS Handbook is to take a more pragmatic view. Thus, not only will the effect of the POSS be examined but also whether it offers an attractive solution compared to those already known or on the market.
Is POSS a Molecule or a Particle?
POSS appear spherical or cubic to the eye and somehow this makes the viewer think that POSS are particles. They are often described in the literature as nanoparticles with all the good and the bad associations that come with that nomenclature. Nano has, in recent years been a buzzword and research on all things nano has been popular and well-funded. On the downside, the term nano now invokes concerns over safety and lack of commercialization means nano has lost much of its shine.
As mentioned earlier, POSS are in fact molecules and that can be proven categorically in several ways. POSS can be analyzed by the following techniques which only apply to molecules:
- Solution NMR spectroscopy (1H or 29Si)
- HPLC
- Mass spectroscopy
- Dielectric spectroscopy
- Some POSS types are liquid at room temperature
- POSS can be melted and recrystallized
- POSS can be recrystallized from solution
POSS dissolves molecularly in solvents if the polarity of the solvent is matched to that of the POSS. The POSS can then be recrystallized from solution to purify it, just as with other solid chemical substances. Suitable solvents for POSS can be predicted using the Solubility Parameter concept, which is also useful for example when one needs to predict the solubility of an as yet unsynthesized POSS type. POSS can be dissolved in solvents in high concentrations, approaching 100 weight percent in some cases. POSS can also be dissolved in polymers but in that case the solubility limit is normally around 5 weight percent.
There are four main types of POSS configuration:
- Eight identical side groups
- A statistical mixture of two different ligands totaling eight
- Seven identical units with one corner of the cage open exposing three silanol groups
- Seven identical groups with one different group inserted in one corner of the cage
So, POSS are molecules that just look reminiscent of particles and that is why they are sometimes referred to as “molicles” to reflect their dual nature. The fact that POSS are molecules is a great advantage because it means they dissolve. In contrast, nanoparticles are notoriously difficult to disperse and that has held back their use in commercial applications. Furthermore, as POSS are not nanoparticles they avoid the safety uncertainties associated with nano materials.
Properties of POSS Polyhedral Oligomeric Silsesquioxanes
Naturally, with well over 200 POSS types in existence, one cannot detail the properties for each and every one. It is more convenient to take an overview (Table 3).
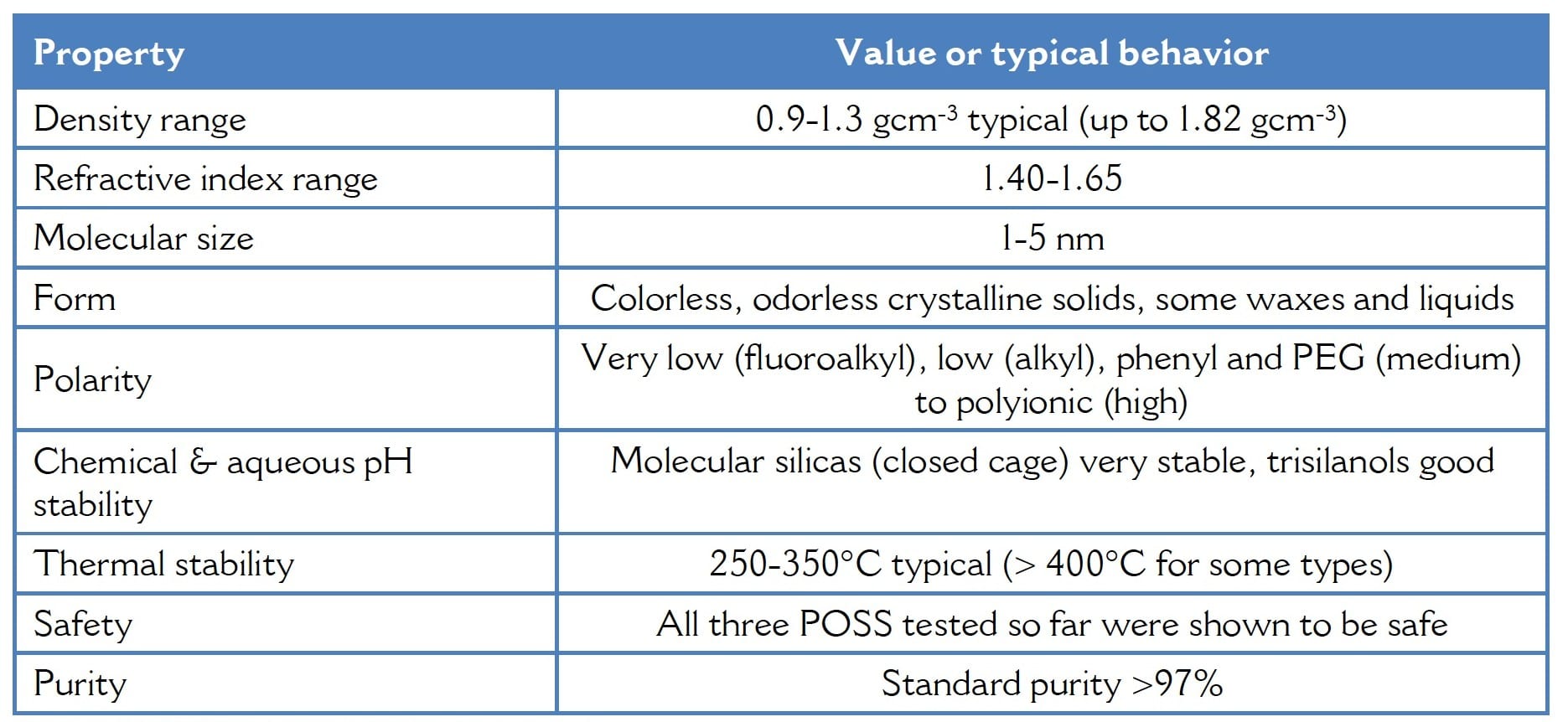
Table 3 POSS Properties Overview
When trying to identify applications for POSS, it is appropriate to start by looking at the unusual and unique properties arising from the POSS molecular structure. These can be summarized as follows where each of the features results in consequences in terms of properties:
- Rigid cage structure
- High thermal stability
- Relatively high molecular weight
- Fully tunable solubility
- Fully tunable reactivity
Special Properties and Applications
In order to have a chance in the marketplace, a product needs to present an attractive value proposition to the customer. Specifically, the following three criteria must be fulfilled:
- The product must produce an effect
- The effect must be useful in some way
- The product must be competitive in terms of cost and performance compared to products incumbent in the marketplace
In order to have a metric of the viability of each proposed application for POSS, these three criteria will be applied in each case. Some applications are mentioned in the section below. The POSS Handbook gives more insight into each possible application, specifically how effective POSS is and whether the application has commercial potential.
POSS Applications
There are many effects that can be achieved with POSS. The ones that will take off are the ones for which POSS is better and / or cheaper than other alternatives. Scientists often look at the effect alone without regard for commercial factors, which is not surprising, because that is not their concern. Here I list some of the applications, several of which I helped develop then assess which ones appear to make commercial sense. More details can be found in the POSS Handbook.
Initially it was thought that POSS could be used to reinforce plastics but that turned out not to be the case. Modulus and strength are not much affected at all. Much later, fluoro POSS was used to make surface very hydrophobic but the same feat can be accomplished at much lower cost using other fluorinated molecules.
POSS flow aids
Small molecules lubricate polymer chains and promote flow. However, they are often thermally unstable and normally they plasticize the polymer, that is modulus and strength both suffer.
POSS has proven useful for enhancing the flow of high-temperature polymers like PEEK, PEI and PPS because of its high thermal stability and the rigid nature of the cage.
The rigid POSS molecule does not plasticize, so mechanical performance i.e. modulus / stiffness is maintained. This was something I helped develop and we were proud to receive the R&D 100 Award for the product range.
It is difficult to find commercial applications for new molecules, especially when they are rather expensive.
Colorless polyimide
Polyimide is normally transparent but has a distinct orange color. By copolymerizing with POSS, the color is removed, thus paving the way for new optical applications. This has been patented and commercialized.
POSS dental restoratives
Dental materials are sold in small amounts and at high prices. Dental glass filler can cost up to $1000 per pound for example. Small amounts of POSS are used in these applications. It is claimed that the POSS molecules are exactly the right size to plug nanotubules in the tooth to prevent sensitivity.
POSS lubricants
The US Air Force looked at some POSS types as potential lubricants but results were disappointing. Years later I led a drive to make and commercialize PEG POSS, a liquid, in bulk. We made it in just 4 weeks and it turned out to be a top selling product. When tested on industry standard equipment, it was found to perform better than the best in class lubricant on the market. Although the PEG POSS is significantly more expensive, it may be that there are niche applications where lubricant performance is paramount and cost is secondary. The other application we targeted was cosmetics and that turned out to be a significant application for PEG POSS.
POSS dispersants
It was been shown that POSS trisilanols, which have one corner of the cage missing to expose highly reactive Si-OH groups, are very effective dispersants, even for particles for which conventional organosilanes do not work. It may be that there are applications, for example in dental materials, where these POSS dispersants would be suitable.
Commercial aspects
Unusual molecules always attract attention from the scientific community but they only become important when they can compete in the real world on availability, price and performance.
In many ways POSS is more attractive than other high profile technologies although still significantly more costly than most plastics additives. The starting material cost for POSS is fixed and rather high so there is a floor on the price of POSS even if it is scaled up.
Conclusion
As we have seen, the POSS family of molecules is interesting and has been the focus of a great deal of research. One or two very niche applications exist already and it can be expected that new applications will gradually be found over the coming decades. Unfortunately, the commercial potential of POSS very much limited in three ways. Firstly, the high cost, which cannot be further lowered because the raw materials used to make POSS are themselves expensive ($10-20 / lb) in bulk for the least expensive types. Secondly, as discussed, the marketing efforts have been focused on applications that make no sense in terms of cost or performance. Lastly, commercial exploitation of new technologies takes in the order of 10-20 years. Based on that last comment we can expect to see applications take off soon.
Further information about POSS
Polyhedral Oligomeric Silsequioxane (POSS) Enhanced Plastics (Chapter 23)
C. DeArmitt, Functional Fillers for Plastics, 2nd Edition, Editor Professor M. Xanthos, Wiley-VCH, 2010.
Polyhedral Oligomeric Silsesquioxanes in Plastics (Chapter 5)
C. DeArmitt, Applications of Polyhedral Oligomeric Silsesquioxanes, Editor C. Hartman-Thompson, Springer Press, 2011.
POSS Handbook
C. DeArmitt (properties and applications of polyhedral oligomericsilsesquioxanes).
POSS User’s Guide
C. DeArmitt POSS User’s Guide 700 downloads per month starting in 2007.
POSS keeps high temperature plastics flowing
C. DeArmitt & P. Wheeler, Plastics Additives & Compounding (Elsevier), 10, 4, 36-39 (2008).
Polyhedral Oligomericsilsequioxanes: Additives for Unique Cosmetic Properties
C. DeArmitt Cosmetics & Toiletries (Allured Publishing), 123, 8, 51-56 (2008).
POSS® is a registered trademark of Hybrid Plastics Inc.
